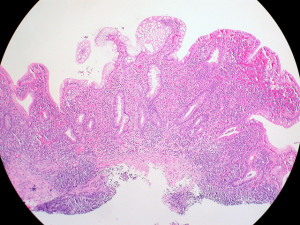
ulcerative colitis
-
Pfizer Expands in Immunology With FDA Approval of New Ulcerative Colitis Drug
Pfizer’s new FDA-approved ulcerative colitis drug Velsipity comes from its $6.7 billion Arena Pharmaceuticals acquisition. The small molecule will compete against blockbuster Bristol Myers Squibb drug Zeposia, which addresses the same target.
-
FDA Rejects Eli Lilly Ulcerative Colitis Drug Over Manufacturing Issues
Rejection of Eli Lilly’s mirikizumab means that two of the four drugs the pharma giant identified as revenue growth drivers for 2023 have failed to pass the regulatory bar. One of those drugs has been approved while the fourth drug still awaits an FDA decision.
-
Jorie Healthcare CEO Shares Why Automation is Critical to Revenue Cycle Management
The revenue cycle management business is using AI tools to automate cumbersome tasks to help hospitals operate more efficiently. It’s beginning to attract the attention of major healthcare organizations.
-
Data and regulatory roundup: Pfizer’s feat, an ADHD miss, Eli Lilly’s CRL & more
A pharmaceutical giant reported a Phase 3 success for a potential blockbuster drug, while four other companies reported disappointing data from their respective clinical trials. Our recap of data readouts and regulatory news also includes one FDA rejection.
-
Pfizer: $6.7B Arena buyout complements rather than replaces JAK drugs
Sales of blockbuster Pfizer drug Xeljanz are under pressure from a stronger safety warning placed across that drug’s entire class. In acquiring Arena Pharmaceuticals for $6.7 billion, Pfizer gets a lead compound with a different and potentially safer approach to treating inflammatory conditions.
-
Despite drug’s Phase 2 failure in ulcerative colitis, BMS keeps blockbuster hopes
Bristol Myers Squibb drug deucravacitinib missed the main and secondary goals of a Phase 2 study in ulcerative colitis. Despite the clinical trial failure, the drug previously posted positive Phase 3 results in plaque psoriasis and BMS still projects the molecule will become a blockbuster seller in multiple autoimmune conditions.
-
Theravance’s Janssen Biotech-partnered ulcerative colitis drug flops in Phase 2
The Theravance Biopharma drug is the lead program in an alliance with Johnson & Johnson subsidiary Janssen Biotech, which paid $100 million up front to develop the JAK inhibitor for intestinal diseases. Following the Phase 2 failure, Theravance said it will minimize spending on that program.
-
Gilead to seek FDA approval for rheumatoid arthritis drug this year
The timing of the filing appears to allay concerns that the FDA would require completion of a study evaluating the testicular safety of the drug, filgotinib, and thus push its launch date from 2020 to 2021-22, an analyst wrote.
-
The expanding ecosystem of microbiome startups
More funding and another powerful pharma backer have entered the microbiome space, showing that at least some investors and pharma execs believe the field can begin converting preclinical ideas into valid human therapies.
-
Navigating Healthcare’s Data Revolution: Priorities, Opportunities, and Challenges for Health Systems
Arcadia recently partnered with HIMSS Market Insights to survey executives, IT, technology, and clinical leaders. Here’s what we found.