Pharma
-
In Same Day, UCB Lands Two FDA Drug Approvals in Autoimmune Diseases
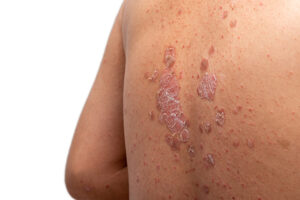
FDA approval of UCB’s Bimzelx gives the Belgian drugmaker a contender in the crowded plaque psoriasis therapies market. In the rare disease generalized myasthenia gravis, the agency approved Zilbrysq, a UCB drug with advantages over two blockbuster AstraZeneca drugs addressing the same target.
-
Thermo Fisher Adds Proteomics to Its Portfolio Through $3.1B Olink Acquisition
Thermo Fisher Scientific is acquiring Olink, a company that provides proteomics analysis tools and services used in drug discovery research. Olink’s growing revenue will help shore up a Thermo Fisher business segment whose sales have fallen due to declining Covid-19 testing demand.
-
Creating an Omnichannel Customer Journey in Healthcare
Improving Customer Experience and Conversion in Healthcare & Life Sciences
-
Roche Deal Aims to Get Molecular Glues to Stick to Elusive Cancer, Neuro Targets
Roche is paying $50 million up front to begin a drug R&D alliance with Monte Rosa Therapeutics, a biotech whose molecular glue technology could address targets previously deemed undruggable. It’s Roche’s second such deal in the past month.
-
What Will Rite Aid Need to Do to Recover from Bankruptcy?
Rite Aid filed for Chapter 11 bankruptcy protection on Sunday amid decreasing sales, billions of dollars in debt and more than a thousand lawsuits claiming the chain filled illegal prescriptions for opioids. In order for the company to get back on its feet, experts say it will have to start acting more like its competitors, such as Walgreens and CVS, by leaning more into care delivery, forging strong payer partnerships, and improving its digital offerings.
-
Pfizer Shaves Revenue Projections, Starts Cost-Cutting Plan Amid Lower Covid-19 Product Demand
Pfizer lowered its revenue projections for 2023, a change it attributed to declining demand for its Covid-19 vaccine, Comirnaty, and antiviral drug, Paxlovid. Citing this revenue decline, Pfizer is implementing a companywide cost-cutting plan projected to save $3.5 billion.
-
Pfizer Expands in Immunology With FDA Approval of New Ulcerative Colitis Drug
Pfizer’s new FDA-approved ulcerative colitis drug Velsipity comes from its $6.7 billion Arena Pharmaceuticals acquisition. The small molecule will compete against blockbuster Bristol Myers Squibb drug Zeposia, which addresses the same target.
-
BioNTech Turns to China Again for Cancer Drugs, Paying $70M to Partner on an ADC
BioNTech gains rights to a MediLink Therapeutics antibody drug conjugate that targets tumors expressing the HER3 protein. The deal comes six months after the German company entered the ADC field by acquiring rights to two therapeutic candidates from DualityBio.
-
Pharma, Artificial Intelligence, BioPharma
Sanofi Adds Biologics to Its AI Ambitions, Striking Up R&D Alliance With BioMap
Formed by Baidu founder and CEO Robin Li, BioMap uses artificial intelligence technology to glean insight into proteins to guide biologic drug discovery. The startup is the latest company to join a growing list of Sanofi partners as the pharmaceutical giant continues investing in AI-enabled drug discovery.
-
Bayer’s New $250M Cell & Gene Therapy Site Signals More Investments to Come
Bayer opens its new cell therapy manufacturing facility as it prepares a Parkinson’s disease cell therapy for Phase 2 testing. The new 100,000 square foot facility also has space for manufacturing other cell therapies in the pharma giant’s pipeline.
-
Why One Expert Thinks the Medicare Drug Price Negotiation Program Is Here to Stay
Merith Basey, executive director of Patients For Affordable Drugs, believes the Medicare price negotiation program is here to stay, no matter who is president and no matter how many Big Pharma players file lawsuits. Her reasoning? Every other high income nation has been negotiating prices with drugmakers for years, and the program is widely popular in the U.S. among pretty much everyone who isn’t a pharma company, she said during a session at Engage at HLTH.
-
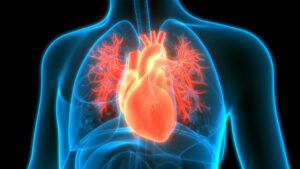
FDA Rejection Is a Delay, Not a Detour for Alnylam’s Aim to Treat Heart Disorder
Despite an affirmative FDA advisory committee vote, the agency declined to approve Alnylam Pharmaceuticals’ Onpattro for treating the heart complications caused by a rare, inherited protein disorder. But Alnylam has other drugs candidates for the disease, including one expected to post Phase 3 data in the first half of 2024.
-
Bristol Myers Squibb Bolsters Its Cancer Presence With $4.8B Mirati Acquisition
Acquiring Mirati Therapeutics brings Bristol Myers Squibb Krazati, one of two FDA-approved therapies addressing a KRAS cancer mutation. The deal comes as BMS looks to add revenue-generating products as patent expirations loom for several of its cancer products.
-
Takeda to Pull Lung Cancer Drug from Market After Failed Confirmatory Study
Takeda Pharmaceutical drug Exkivity failed the confirmatory study required of its 2021 accelerated approval. Our recap of other recent regulatory developments includes a partial clinical hold on a cancer drug, a Covid-19 vaccine authorization, and several drug approvals in the U.S. and beyond.